Findings indicate broader implications towards establishment of immune competency and priming before birth, a concept not explored before in fetal immunity
Using a combination of complementary approaches, Mishra et al identified the presence of live microbes in healthy human fetal tissues that prime T-cell during second trimester of gestation.
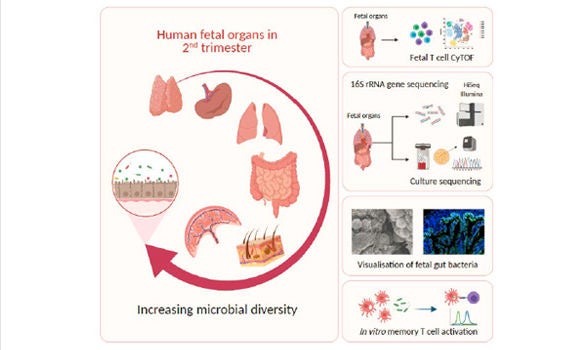
2 June 2021, Singapore – The human fetal immune system begins to develop early during gestation, however, factors responsible for fetal immune-priming remain elusive. Using multiple complementary approaches, Dr Florent Ginhoux from A*STAR’s Singapore Immunology Network (SIgN), Professor Jerry Chan from KK Women’s and Children’s Hospital (KKH), Professor Salvatore Albani from the SingHealth Duke-NUS Translational Immunology Institute, with collaborators from Cambridge University explored potential exposure to microbial agents in-utero. The team identified live microbes across fetal organs that stimulate activation of fetal T-cells during the second trimester of gestation. The study was published in the scientific journal, Cell on 24 June 2021.
The team profiled microbes across fetal organs using 16S-rRNA gene sequencing and detected low but consistent microbial signal in fetal gut, skin, placenta and lungs, in the second trimester of gestation. They identified several live bacterial strains including Staphylococcus and Lactobacillus in fetal tissues, which induced in vitro activation of memory T-cells in fetal mesenteric lymph-node, supporting the role of microbial exposure in fetal immune-priming. Finally, using scanning electron microscope (SEM) and Ribonucleic acid (RNA) in situ hybridisation (RNA-ISH), discrete localisation of bacteria-like structures and eubacterial-RNA were visualised within the 14th week fetal gut lumen. These findings indicate selective presence of live microbes in fetal organs during second trimester of gestation and have broader implications on the establishment of immune competency and priming before birth.
The findings demonstrate that healthy human fetal tissues (in the second trimester of gestation) contain effector memory T-cells, a sparse biomass of bacteria and an active memory T-cell response towards fetal bacteria. It also demonstrates direct spatial localisation of bacterial entities, localised within the lumen of developing fetal gut, during second trimester of gestation.
Dr Florent Ginhoux, Senior Principal Investigator, SIgN and co-last author of the study said, “Our study demonstrates that such microbial presence primes the fetal immune system, thereby putting early microbial memory in the context of fetal immune priming, a concept not explored before in fetal immunity. It will be interesting to explore the precise nature of these microbial antigen-specific circulatory and immune cells that reside in human fetal organ tissues, and their potential role in imparting selective defence against pathogenic microbes in neonatal and adult life. Taken together, these findings have wider implications in understanding the key factors involved in fetal immune system development and priming in utero, which may set the basis for life-long human health and immunity of the organism.”
Co-last author of the study, Professor Jerry Chan, Senior Consultant, Department of Reproductive Medicine, KK Women’s and Children’s Hospital (KKH), and Senior National Medical Research Council Clinician Scientist said, “This study shows us that the vertical transmission of microbial organisms from mother to child through a yet unknown mechanism may have important implications in imparting influences such as immune education or priming to the baby in both immunity (including auto-immunity) and tolerance in later life. From a clinical standpoint, this finding adds to our understanding of how the fetus or newborn may respond to infections or intrauterine stem cell transplantation.”
Co-senior author of the study, Professor Salvatore Albani, Director of the SingHealth Duke-NUS Translational Immunology Institute and Professor of Medicine at Duke-NUS said, “This study is a paramount example of a multidisciplinary, truly translational approach to improve our understanding of how our immune system develops and relates to the environment. It shatters the notion of the fetus living in an isolated, privileged immune environment and emphasises the importance of the maternal-fetal interface in the context of environmental challenges and experiences. The research approach taken relies on high dimensionality techniques which enable the depiction and the dissection of the fetal Immunome systematically. This research bears important immediate translational implications as it will affect current and future approaches to manipulation of the immune system in prenatal and perinatal age.”
Co-last author of the study, Naomi McGovern, PhD, Department of Pathology, University of Cambridge, UK, said: “This study provides valuable insight into fetal immunology and demonstrates that complex immune priming events occur during gestation. It shows that fetal dendritic cells are sensitive to diverse signaling cues and can initiate appropriate responses that are required for healthy fetal development.“
More information on the study, “Microbial exposure during early human development primes fetal immune cells” can be found via the team’s published paper in Cell:
https://doi.org/10.1016/j.cell.2021.04.039












