Since the start of the COVID‐19 pandemic, KK Women’s and Children’s Hospital (KKH) has been at the frontline of screening and treating pregnant women suspected or confirmed to have COVID‐19.
Majority of pregnant women admitted to hospital with COVID-19 are unvaccinated
From May to September 2021, KKH saw a significant increase in the number of pregnant women infected with COVID‐19 being admitted, compared to the same period last year. Although the majority experienced mild symptoms, some developed complications requiring oxygen support and close monitoring to prevent or treat complications such as pneumonia or thrombosis (blood clots). The average length of hospital stay for these unvaccinated pregnant women was two to three weeks.
It is worrying that in Singapore, United Kingdom and United States, the majority of the pregnant women infected with COVID‐19 admitted to hospital, are not vaccinated. Worldwide, an increasing number of pregnant women are being infected with COVID‐19 and dying from it. Compared to the non‐pregnant population, pregnant women are at higher risk of severe complications if they are infected with the virus. They also have double the risk of preterm birth if they are symptomatic, and their babies may require neonatal intensive care.
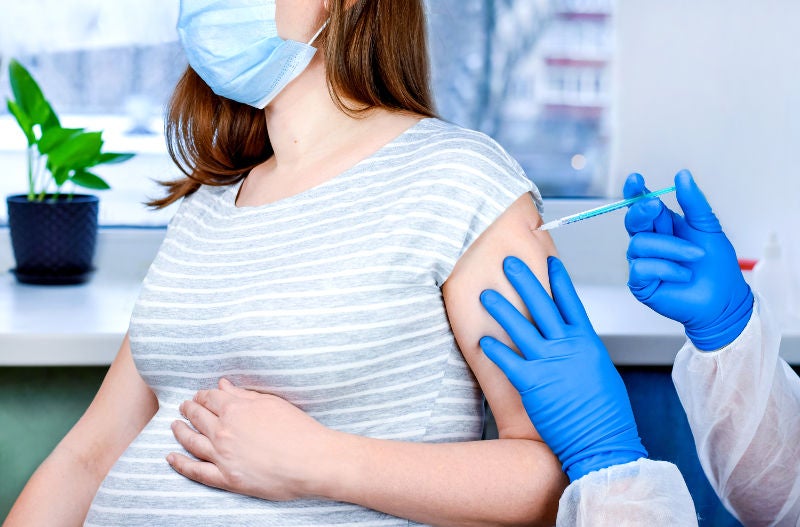
COVID-19 vaccination is safe and effectively protects mother and baby
Current evidence shows that COVID‐19 vaccination is safe and protects the mother and baby. The vaccines have been shown to generate antibody production in pregnant women with passive transfer to the baby antenatally or before birth. As Singapore moves towards COVID‐resilience, it is strongly recommended that all pregnant women or women intending to conceive, receive the COVID‐19 vaccination.
- A fully vaccinated pregnant woman who contracts COVID-19 is more likely to have no or mild symptoms only.
- Vaccinated women are less likely to need an emergency or early delivery for COVID-19-related complications. As at September 2021, no vaccinated pregnant KKH patient who contracted COVID-19 has needed oxygen therapy or intensive care support.
- COVID-19 antibodies from vaccinated mothers can be transferred to babies via the placenta or breast milk. Hence, fully vaccinated pregnant women are recommended to have the booster dose, to boost or maintain antibody levels.
Vaccination is safe at all stages of pregnancy
The COVID-19 vaccination can be safely administered at all stages of pregnancy, and does not change the DNA of the individual or the mode of delivery. There is no evidence that COVID-19 vaccination causes miscarriages or birth defects. A 2021 study has shown no increase in birth defects or disorders among babies born to mothers vaccinated against COVID-19, compared to babies born before the pandemic.
In the United States, nearly 170,000 pregnant women have been vaccinated – as of October 2021 (using Pfizer BioNTech or Moderna vaccines) and no safety concerns have been identified. To reap the full benefits, vaccination early in pregnancy is recommended to allow time for antibodies to develop and sufficient quantities to pass through the placenta into the baby before birth.
COVID-19 in women can affect the fetus and newborn.
Strong evidence shows that COVID-19 can be potentially transmitted from mother to baby through respiratory droplets, opportunistic aerosol transmission and contact/surface transmission. Rare case reports have been published of trans-placental transmission.
COVID-19 risks to a newborn can include complications during pregnancy such as prematurity, low birth weight, fetal distress and stillbirth. After birth, moderate and severe complications in an infected newborn include pneumonia, respiratory distress, vomiting, diarrhoea, occasionally low blood counts and collapsed lungs.
Protection in a COVID-19-endemic world
As COVID-19 becomes endemic in countries around the world, to protect themselves and their babies against the risk of severe disease, all pregnant women or women intending to conceive are recommended to receive the COVID‐19 vaccination.
Read more |
 | Professor Tan Hak Koon, Chairman, Division of Obstetrics and Gynaecology, KKH Prof Tan Hak Koon is Chairman and Senior Consultant with the Division of Obstetrics and Gynaecology at KKH. At SingHealth, he holds the appointment of Chairman of the SingHealth-Duke NUS Obstetrics and Gynaecology Academic Clinical Programme (OBGYN-ACP) and Designated Institutional Official (DIO) at SingHealth Residency concurrently. Additionally, he is an Associate Dean, Office of Academic and Clinical Development, Duke-NUS Medical School. |
References:
|













